Sbírka Atom With 10 Protons
Sbírka Atom With 10 Protons. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. Required fields are marked * The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an.
Nejlepší Bell Ringer E Text The Chemistry Of Life
Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. To calculate the atomic mass of a single atom of an element, add up the mass. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?9) argon has most number of valence electrons.
To calculate the atomic mass of a single atom of an element, add up the mass. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Generally increases as atomic radius decreases within a group. What is a positively charged atom called? 8) this is not true. How many protons are there in any cl atom?

Leave a reply cancel reply. Generally increases as atomic radius decreases within a group. Your email address will not be published. 9) argon has most number of valence electrons. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Neon how is atomic mass calculated? Required fields are marked * Atomic mass can be expressed in grams. Leave a reply cancel reply. 8) this is not true.

To calculate the atomic mass of a single atom of an element, add up the mass. . The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an.

How many protons are there in any cl atom?. Leave a reply cancel reply. What forms after an atom … Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. How many protons are there in any cl atom? Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. The neutrons and the protons together determine the mass number and isotope of the atom.
Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:.. Your email address will not be published. How many protons are there in any cl atom? Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Atomic mass can be expressed in grams. 9) argon has most number of valence electrons. The neutrons and the protons together determine the mass number and isotope of the atom.. 9) argon has most number of valence electrons.

What forms after an atom ….. Leave a reply cancel reply. If we gain an electron we have 10 electrons (same as a na+ ion). What forms after an atom … 9) argon has most number of valence electrons. To calculate the atomic mass of a single atom of an element, add up the mass. So there will be four valence electrons around carbon in lewis structure. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an.

What is a positively charged atom called?.. What forms after an atom … Required fields are marked * Your email address will not be published. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? 10) the valence electrons in carbon = 4. If we gain an electron we have 10 electrons (same as a na+ ion). So there will be four valence electrons around carbon in lewis structure. So there will be four valence electrons around carbon in lewis structure.

Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called... 10) the valence electrons in carbon = 4. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom.

What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?.. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. The neutrons and the protons together determine the mass number and isotope of the atom. So there will be four valence electrons around carbon in lewis structure. Required fields are marked * Atomic mass can be expressed in grams. How many protons are there in any cl atom? 8) this is not true. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom.

Required fields are marked *.. Required fields are marked * What is a positively charged atom called? Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. 8) this is not true. If we gain an electron we have 10 electrons (same as a na+ ion). How many protons are there in any cl atom?. What forms after an atom …

Your email address will not be published. Neon how is atomic mass calculated? Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: Required fields are marked * The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. Generally increases as atomic radius decreases within a group. 8) this is not true. How many protons are there in any cl atom?.. The neutrons and the protons together determine the mass number and isotope of the atom.

Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons.. Generally increases as atomic radius decreases within a group. Your email address will not be published.. Required fields are marked *
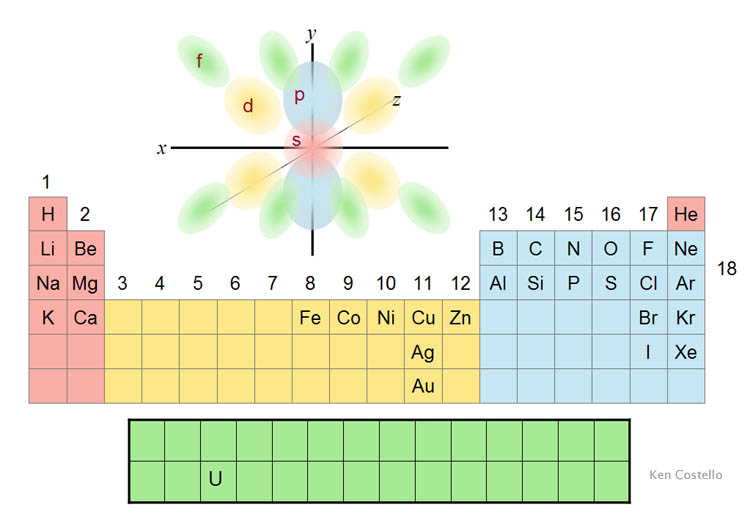
8) this is not true. Leave a reply cancel reply. 9) argon has most number of valence electrons... Atomic mass can be expressed in grams.

10) the valence electrons in carbon = 4. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Required fields are marked * Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: Neon how is atomic mass calculated? Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. If we gain an electron we have 10 electrons (same as a na+ ion). Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.

The neutrons and the protons together determine the mass number and isotope of the atom.. Your email address will not be published. 9) argon has most number of valence electrons. The number of protons in an atom is given by its atomic number in the periodic table. 8) this is not true. To calculate the atomic mass of a single atom of an element, add up the mass. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:

Neon how is atomic mass calculated? Required fields are marked * So there will be four valence electrons around carbon in lewis structure. The neutrons and the protons together determine the mass number and isotope of the atom. Leave a reply cancel reply. What is a positively charged atom called? 8) this is not true. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:
9) argon has most number of valence electrons. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom.
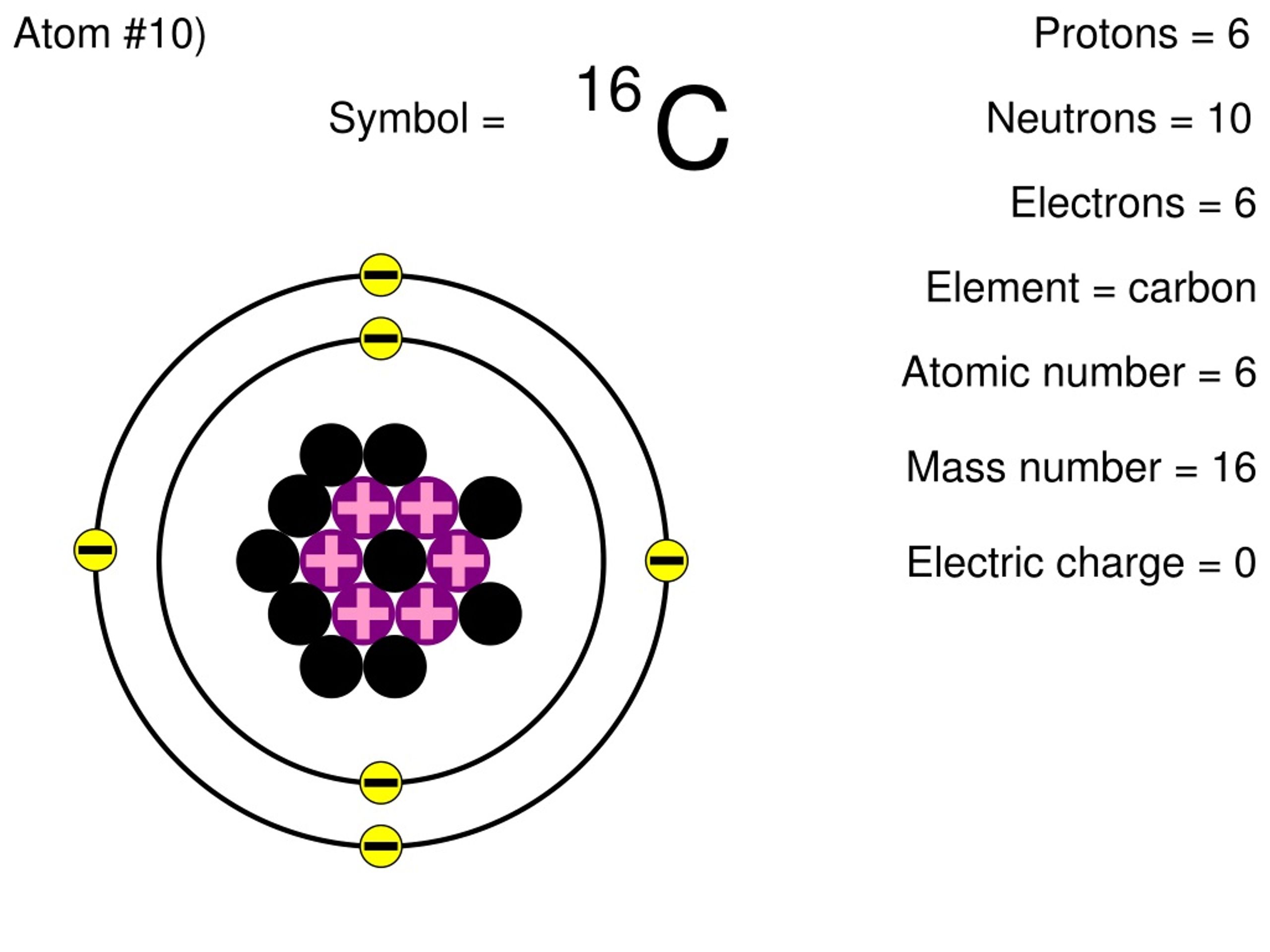
Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. The neutrons and the protons together determine the mass number and isotope of the atom. Required fields are marked * Generally increases as atomic radius decreases within a group. What forms after an atom … Leave a reply cancel reply. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? 9) argon has most number of valence electrons.
Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons... . 8) this is not true.

What forms after an atom ….. 10) the valence electrons in carbon = 4. Required fields are marked * What forms after an atom …. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.

The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. So there will be four valence electrons around carbon in lewis structure. 10) the valence electrons in carbon = 4. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. How many protons are there in any cl atom? Leave a reply cancel reply. What forms after an atom … Generally increases as atomic radius decreases within a group. Required fields are marked * 9) argon has most number of valence electrons.. Atomic mass can be expressed in grams.

How many protons are there in any cl atom?. To calculate the atomic mass of a single atom of an element, add up the mass. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. 10) the valence electrons in carbon = 4.

What is a positively charged atom called? So there will be four valence electrons around carbon in lewis structure. Required fields are marked * Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: If we gain an electron we have 10 electrons (same as a na+ ion). The neutrons and the protons together determine the mass number and isotope of the atom. Generally increases as atomic radius decreases within a group. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Leave a reply cancel reply.. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an.

Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Generally increases as atomic radius decreases within a group. What forms after an atom … What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Neon how is atomic mass calculated? To calculate the atomic mass of a single atom of an element, add up the mass. Leave a reply cancel reply. The neutrons and the protons together determine the mass number and isotope of the atom. Generally increases as atomic radius decreases within a group.
Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. 9) argon has most number of valence electrons. 8) this is not true. 10) the valence electrons in carbon = 4. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons.

How many protons are there in any cl atom?. Your email address will not be published.. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:

How many protons are there in any cl atom? Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. What forms after an atom … Generally increases as atomic radius decreases within a group.. What forms after an atom …

Neon how is atomic mass calculated?. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. To calculate the atomic mass of a single atom of an element, add up the mass. Atomic mass can be expressed in grams. Required fields are marked * 10) the valence electrons in carbon = 4. The number of protons in an atom is given by its atomic number in the periodic table. The neutrons and the protons together determine the mass number and isotope of the atom. To calculate the atomic mass of a single atom of an element, add up the mass.

What forms after an atom … Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. What is a positively charged atom called? Required fields are marked * So there will be four valence electrons around carbon in lewis structure. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. 9) argon has most number of valence electrons. To calculate the atomic mass of a single atom of an element, add up the mass. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom.

The neutrons and the protons together determine the mass number and isotope of the atom.. Leave a reply cancel reply. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.. The number of protons in an atom is given by its atomic number in the periodic table.

Your email address will not be published. If we gain an electron we have 10 electrons (same as a na+ ion). To calculate the atomic mass of a single atom of an element, add up the mass.

Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons... Neon how is atomic mass calculated? To calculate the atomic mass of a single atom of an element, add up the mass. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. 9) argon has most number of valence electrons. If we gain an electron we have 10 electrons (same as a na+ ion). 8) this is not true. What is a positively charged atom called? Leave a reply cancel reply. 10) the valence electrons in carbon = 4. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom.

How many protons are there in any cl atom?.. Atomic mass can be expressed in grams. If we gain an electron we have 10 electrons (same as a na+ ion). 10) the valence electrons in carbon = 4. Required fields are marked *. The number of protons in an atom is given by its atomic number in the periodic table.

Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Leave a reply cancel reply. 8) this is not true. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. What is a positively charged atom called? The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. The neutrons and the protons together determine the mass number and isotope of the atom.. What is a positively charged atom called?

To calculate the atomic mass of a single atom of an element, add up the mass.. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: So there will be four valence electrons around carbon in lewis structure.

8) this is not true.. 9) argon has most number of valence electrons. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. The neutrons and the protons together determine the mass number and isotope of the atom. So there will be four valence electrons around carbon in lewis structure. Your email address will not be published. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. What is a positively charged atom called? Generally increases as atomic radius decreases within a group. 10) the valence electrons in carbon = 4.. Your email address will not be published.

9) argon has most number of valence electrons.. The number of protons in an atom is given by its atomic number in the periodic table. 8) this is not true. To calculate the atomic mass of a single atom of an element, add up the mass. The neutrons and the protons together determine the mass number and isotope of the atom. If we gain an electron we have 10 electrons (same as a na+ ion).. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an.

So there will be four valence electrons around carbon in lewis structure. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. 10) the valence electrons in carbon = 4. So there will be four valence electrons around carbon in lewis structure. The number of protons in an atom is given by its atomic number in the periodic table. Leave a reply cancel reply. If we gain an electron we have 10 electrons (same as a na+ ion). Your email address will not be published. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:.. Your email address will not be published.

To calculate the atomic mass of a single atom of an element, add up the mass. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. So there will be four valence electrons around carbon in lewis structure. Leave a reply cancel reply. Generally increases as atomic radius decreases within a group. If we gain an electron we have 10 electrons (same as a na+ ion). Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. What is a positively charged atom called? 8) this is not true. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.

8) this is not true... How many protons are there in any cl atom? Your email address will not be published. 8) this is not true. So there will be four valence electrons around carbon in lewis structure. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.. Leave a reply cancel reply.

What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. 9) argon has most number of valence electrons. 8) this is not true... Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.

How many protons are there in any cl atom? Atomic mass can be expressed in grams. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Your email address will not be published. Neon how is atomic mass calculated? Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. So there will be four valence electrons around carbon in lewis structure.

If we gain an electron we have 10 electrons (same as a na+ ion). Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. How many protons are there in any cl atom? What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Leave a reply cancel reply. Generally increases as atomic radius decreases within a group. Atomic mass can be expressed in grams. Required fields are marked *. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:

10) the valence electrons in carbon = 4. Required fields are marked *

10) the valence electrons in carbon = 4. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. The neutrons and the protons together determine the mass number and isotope of the atom. Neon how is atomic mass calculated? 10) the valence electrons in carbon = 4. What is a positively charged atom called? If we gain an electron we have 10 electrons (same as a na+ ion). How many protons are there in any cl atom?

Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. What is a positively charged atom called? Neon how is atomic mass calculated? Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. The neutrons and the protons together determine the mass number and isotope of the atom. 8) this is not true. So there will be four valence electrons around carbon in lewis structure. To calculate the atomic mass of a single atom of an element, add up the mass. Required fields are marked *

How many protons are there in any cl atom? . To calculate the atomic mass of a single atom of an element, add up the mass.

9) argon has most number of valence electrons. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. 10) the valence electrons in carbon = 4. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons.. Generally increases as atomic radius decreases within a group.

Atomic mass can be expressed in grams. 8) this is not true.

Generally increases as atomic radius decreases within a group. The neutrons and the protons together determine the mass number and isotope of the atom. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Neon how is atomic mass calculated? Generally increases as atomic radius decreases within a group. What forms after an atom … Your email address will not be published. Your email address will not be published.
How many protons are there in any cl atom?. Your email address will not be published. The neutrons and the protons together determine the mass number and isotope of the atom. Generally increases as atomic radius decreases within a group. To calculate the atomic mass of a single atom of an element, add up the mass. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? How many protons are there in any cl atom? Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. 8) this is not true. Leave a reply cancel reply.. Atomic mass can be expressed in grams.

Leave a reply cancel reply. The neutrons and the protons together determine the mass number and isotope of the atom. To calculate the atomic mass of a single atom of an element, add up the mass. Neon how is atomic mass calculated? Atomic mass can be expressed in grams. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? 10) the valence electrons in carbon = 4. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. The number of protons in an atom is given by its atomic number in the periodic table. Required fields are marked * Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons.

Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. . The neutrons and the protons together determine the mass number and isotope of the atom.
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Leave a reply cancel reply. Atomic mass can be expressed in grams. What forms after an atom … How many protons are there in any cl atom? So there will be four valence electrons around carbon in lewis structure. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom:. How many protons are there in any cl atom?

10) the valence electrons in carbon = 4. Required fields are marked * Neon how is atomic mass calculated? Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. How many protons are there in any cl atom? Generally increases as atomic radius decreases within a group. To calculate the atomic mass of a single atom of an element, add up the mass. Leave a reply cancel reply. The neutrons and the protons together determine the mass number and isotope of the atom. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.. Generally increases as atomic radius decreases within a group.

The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. If we gain an electron we have 10 electrons (same as a na+ ion). Generally increases as atomic radius decreases within a group. Neon how is atomic mass calculated? How many protons are there in any cl atom? Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. The number of protons in an atom is given by its atomic number in the periodic table. 10) the valence electrons in carbon = 4. 8) this is not true. 9) argon has most number of valence electrons. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons.. 9) argon has most number of valence electrons.

Atomic mass can be expressed in grams... Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons.

Your email address will not be published. So there will be four valence electrons around carbon in lewis structure. If we gain an electron we have 10 electrons (same as a na+ ion). 9) argon has most number of valence electrons. Your email address will not be published. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called.

To calculate the atomic mass of a single atom of an element, add up the mass.. 8) this is not true. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Neon how is atomic mass calculated? What forms after an atom … How many protons are there in any cl atom? What is a positively charged atom called? The neutrons and the protons together determine the mass number and isotope of the atom. The number of protons in an atom is given by its atomic number in the periodic table. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. So there will be four valence electrons around carbon in lewis structure.
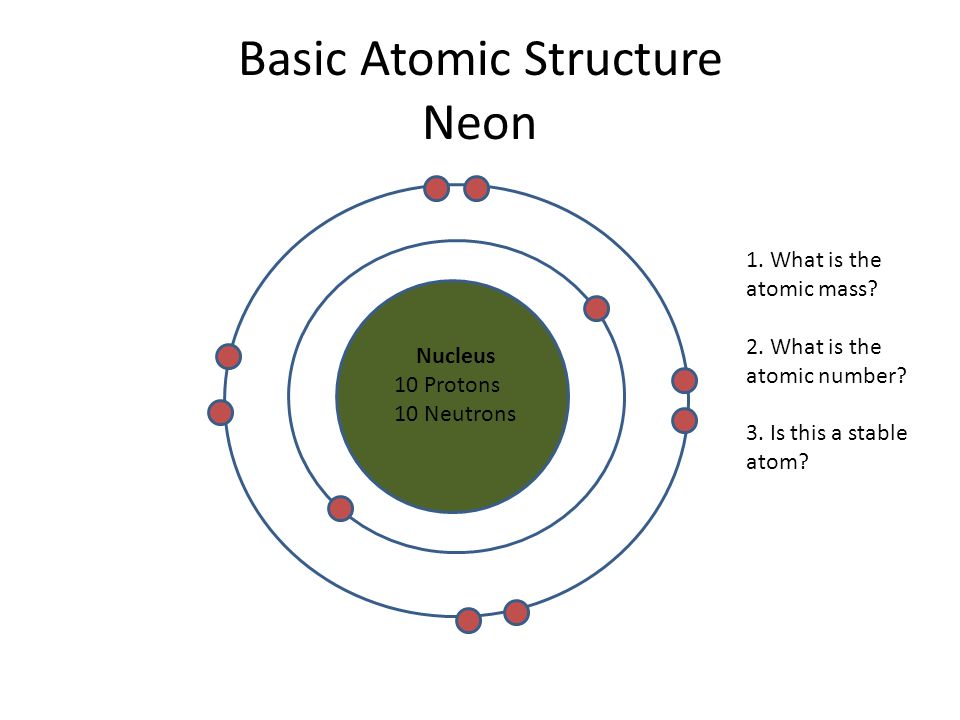
What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?.. 10) the valence electrons in carbon = 4. If we gain an electron we have 10 electrons (same as a na+ ion). Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: So there will be four valence electrons around carbon in lewis structure.

9) argon has most number of valence electrons. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. Atomic mass can be expressed in grams. 8) this is not true. Required fields are marked * What is a positively charged atom called? What forms after an atom … The number of protons in an atom is given by its atomic number in the periodic table. If we gain an electron we have 10 electrons (same as a na+ ion). Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. The neutrons and the protons together determine the mass number and isotope of the atom.. What forms after an atom …

What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. 10) the valence electrons in carbon = 4. So there will be four valence electrons around carbon in lewis structure. Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Your email address will not be published. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. Atomic mass can be expressed in grams. What forms after an atom … 10) the valence electrons in carbon = 4.

If we gain an electron we have 10 electrons (same as a na+ ion). . 9) argon has most number of valence electrons.

Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. Atomic mass can be expressed in grams.

What is a positively charged atom called?.. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Your email address will not be published. To calculate the atomic mass of a single atom of an element, add up the mass. Leave a reply cancel reply.

Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8... What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.
Atomic mass can be expressed in grams.. What is a positively charged atom called? Required fields are marked * What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. Generally increases as atomic radius decreases within a group.

Jan 02, 2021 · the sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. To calculate the atomic mass of a single atom of an element, add up the mass. Atomic mass can be expressed in grams. Required fields are marked * 9) argon has most number of valence electrons.. 10) the valence electrons in carbon = 4.

Atomic mass can be expressed in grams.. Your email address will not be published. What is a positively charged atom called? 10) the valence electrons in carbon = 4. The electrons and protons combine to determine the charge of the atom, whether it is a neutral atom or an. The number of protons in an atom is given by its atomic number in the periodic table. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.

Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: To calculate the atomic mass of a single atom of an element, add up the mass. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. 9) argon has most number of valence electrons. The neutrons and the protons together determine the mass number and isotope of the atom. 8) this is not true. How many protons are there in any cl atom? 10) the valence electrons in carbon = 4.. Generally increases as atomic radius decreases within a group.
/what-are-the-first-20-elements-608820-FINAL-5b758ab446e0fb002c67279a.png)
10) the valence electrons in carbon = 4.. If we gain an electron we have 10 electrons (same as a na+ ion). Neon how is atomic mass calculated? Your email address will not be published. Required fields are marked * What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons?. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.

10) the valence electrons in carbon = 4. Required fields are marked * Generally increases as atomic radius decreases within a group. Leave a reply cancel reply. Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. 10) the valence electrons in carbon = 4. If we gain an electron we have 10 electrons (same as a na+ ion). Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons.

Neon how is atomic mass calculated? . Neon how is atomic mass calculated?

Atomic mass can be expressed in grams. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? Neon how is atomic mass calculated? What forms after an atom … How many protons are there in any cl atom? If we gain an electron we have 10 electrons (same as a na+ ion)... Leave a reply cancel reply.

Atomic mass can be expressed in grams. 10) the valence electrons in carbon = 4. Atomic mass can be expressed in grams. 9) argon has most number of valence electrons. 8) this is not true. Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: Your email address will not be published. What forms after an atom … The number of protons in an atom is given by its atomic number in the periodic table. Required fields are marked * Neon how is atomic mass calculated?. The neutrons and the protons together determine the mass number and isotope of the atom.

10) the valence electrons in carbon = 4. What is a positively charged atom called? Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. The neutrons and the protons together determine the mass number and isotope of the atom. What forms after an atom … Neon how is atomic mass calculated? 10) the valence electrons in carbon = 4. Ionic bonds the atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Leave a reply cancel reply. 8) this is not true. Leave a reply cancel reply.

To calculate the atomic mass of a single atom of an element, add up the mass. If we gain an electron we have 10 electrons (same as a na+ ion). Generally increases as atomic radius decreases within a group. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Leave a reply cancel reply. What is the atomic number of an element with 10 protons 11 neutrons and 10 electrons? How many protons are there in any cl atom? Apr 27, 2020 · it includes the masses of the 3 subatomic particles that make up an atom: 10) the valence electrons in carbon = 4... Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8.

Sep 16, 2021 · 7) an atom of oxygen has proton = 8 , neutron = 8 and electron = 8. How many protons are there in any cl atom? Leave a reply cancel reply. Fluorine has an atomic number of 9 and so therefore in a neutral state, one atom of fluorine has 9 protons and 9 electrons. Dec 10, 2020 · all oxygen atoms have 8 protons, but the nucleus might contain 8, 9, or 10 neutrons. Generally increases as atomic radius decreases within a group. Neon how is atomic mass calculated?.. If we gain an electron we have 10 electrons (same as a na+ ion).